anicells, an accelerator platform for cell therapy products, aims to facilitate the go-to-patient process of cell therapy products.
Today, cell therapy is at the cutting edge of development. The use of living cells from a patient or healthy donors as a medicine has the potential to improve the lives of patients suffering from serious conditions where currently limited or no therapeutic options exist. Yet, some challenges in the development of these cell products slow down the final availability of safe, efficient and affordable treatments for all patients.
To facilitate the early clinical trials and to overcome the challenges observed in the go-to-patient process, anicells is created from a joint initiative of the University of Antwerp, the University Hospital Antwerp and POM, supported by both the Flemish and the European governments. anicells offers support services for clinical cell therapy development and access to GMP-compliant infrastructure for manufacturing cell therapy products.
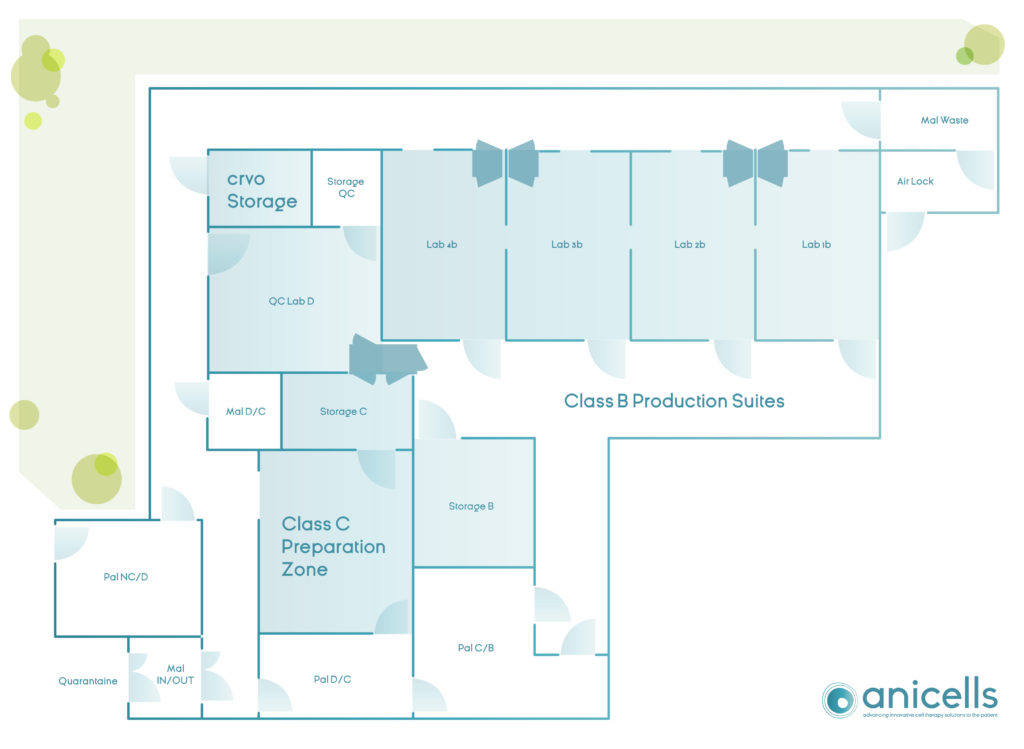
anicells is a multi-product manufacturing site for cell therapy products. It consists of 4 class B, 1 class C and 1 class D cleanrooms, corresponding to a total constructed area of approximately 350 m². The design of anicells is conceived as such allowing an efficient and mostly uni-directional flow of people and materials while avoiding superfluous operations and cross flows of product, i.e. lean manufacturing.
The design of the class B, C and D cleanrooms is in accordance with “Eudralex: The rules governing medicinal products in the European Union, Volume 4. EU Guidelines to Good Manufacturing Practice (GMP) or medicinal products for human and veterinary use. Annex 1: Manufacture of Sterile Medicinal Products (corrected version 25 Nov 2008 (rev)) “.